In labs, classrooms and industrial setups, some chemical changes pull heat in rather than give it off, altering temperatures and driving processes from cooling packs to distillation. Observing which reactions absorb energy helps you understand thermal management, reaction feasibility and everyday phenomena like evaporation.
There are 20 Endothermic Reactions, ranging from Decomposition of ammonium chloride (to gases) to Vaporization of water. For each entry you’ll find below the Equation,ΔH (kJ/mol),Typical conditions / Where found so you can compare heat uptake and real-world context — you’ll find the full list and data below.
How do endothermic reactions differ from exothermic ones?
Endothermic processes absorb heat from their surroundings, so the system’s enthalpy change (ΔH) is positive and the surroundings cool; common examples include melting, vaporization and certain decompositions. Exothermic reactions release heat (negative ΔH) and warm their surroundings, so checking ΔH and measuring temperature change quickly shows which type you have.
Can I observe an endothermic reaction safely at home or in class?
Yes — simple demos like dissolving ammonium nitrate in water or watching ice melt with controlled heating show heat absorption, but always use goggles, gloves and proper containers, avoid ingesting chemicals, work in a ventilated area, and follow teacher or lab-safety instructions; measure temperature with a probe or thermometer to document the effect.
Endothermic Reactions
| Name | Equation | ΔH (kJ/mol) | Typical conditions / Where found |
|---|---|---|---|
| Melting of ice | H2O(s) -> H2O(l) | 6.01 | 0 °C; everyday freezing/melting |
| Vaporization of water | H2O(l) -> H2O(g) | 40.65 | Boiling/evaporation at 100 °C or ambient evaporation |
| Sublimation of carbon dioxide (dry ice) | CO2(s) -> CO2(g) | 25.20 | Below −78.5 °C; dry ice in cooling/transport |
| Sublimation of iodine | I2(s) -> I2(g) | 62.40 | Room temperature to warm; laboratory sublimation |
| Dissolution of ammonium nitrate | NH4NO3(s) -> NH4+(aq) + NO3-(aq) | 25.70 | Room temperature; cold packs, laboratory |
| Dissolution of potassium nitrate | KNO3(s) -> K+(aq) + NO3-(aq) | 34.90 | Aqueous solutions; fertilizer chemistry |
| Dissolution of potassium chloride | KCl(s) -> K+(aq) + Cl-(aq) | 17.22 | Aqueous solutions; labs, physiology buffers |
| Dissolution of ammonium chloride | NH4Cl(s) -> NH4+(aq) + Cl-(aq) | 14.80 | Aqueous solutions; cold baths, labs |
| Vaporization of ethanol | C2H5OH(l) -> C2H5OH(g) | 38.56 | Near room temperature to 78 °C; solvents, fuels |
| Dissociation of dinitrogen tetroxide | N2O4(g) -> 2 NO2(g) | 57.20 | Gas phase; equilibrium at ambient temperatures |
| Formation of ozone | 3/2 O2(g) -> O3(g) | 142.70 | High-energy/UV environments; upper atmosphere |
| Formation of nitric oxide | N2(g) + O2(g) -> 2 NO(g) | 180.60 | High temperature combustion, lightning, engines |
| Electrolysis of water | 2 H2O(l) -> 2 H2(g) + O2(g) | 571.60 | Electrochemical cells; industrial hydrogen production |
| Photosynthesis (overall) | 6 CO2(g) + 6 H2O(l) -> C6H12O6(s) + 6 O2(g) | 2,870 | In plants with sunlight; biosphere energy capture |
| Decomposition of calcium carbonate | CaCO3(s) -> CaO(s) + CO2(g) | 178.30 | High temperature; lime production, calcination |
| Decomposition of ammonium chloride (to gases) | NH4Cl(s) -> NH3(g) + HCl(g) | 176.20 | Heating solid; sublimation/decomposition in labs |
| Thermal decomposition of sodium bicarbonate | 2 NaHCO3(s) -> Na2CO3(s) + CO2(g) + H2O(g) | 129.50 | Heating (baking); oven temperatures |
| Decomposition of magnesium carbonate | MgCO3(s) -> MgO(s) + CO2(g) | 118.00 | High temperature; mineral processing, labs |
| Steam reforming of methane | CH4(g) + H2O(g) -> CO(g) + 3 H2(g) | 206.00 | 700–1,100 °C; industrial hydrogen production |
| Decomposition of dinitrogen pentoxide | 2 N2O5(g) -> 4 NO2(g) + O2(g) | 110.00 | Warming/dissociation; laboratory and atmospheric |
Images and Descriptions

Melting of ice
Ice melting absorbs heat from the surroundings as solid water becomes liquid. It’s familiar in daily life, key to climate processes and refrigeration, and a clear example of a small endothermic phase change per mole of H2O.

Vaporization of water
Turning liquid water into steam requires substantial heat (latent heat of vaporization). This endothermic step cools skin during sweating and is central to distillation, weather, and steam-based power cycles.

Sublimation of carbon dioxide (dry ice)
Dry ice sublimates directly from solid to gas, absorbing heat without melting. That endothermic change makes it useful for refrigeration, theatrical fog effects and controlled cooling where liquid mess must be avoided.

Sublimation of iodine
Iodine crystals vaporize to a purple gas on gentle heating, an endothermic transition. It’s a classic lab demonstration, used historically for purification and visualization of sublimation behavior.

Dissolution of ammonium nitrate
Ammonium nitrate dissolves endothermically, pulling heat from surroundings and producing cold. This property powers disposable instant cold packs and illustrates how solvation and lattice energies control solution enthalpy.

Dissolution of potassium nitrate
Potassium nitrate absorbs heat on dissolving, causing solution cooling. It’s relevant to fertilizer manufacture and classic thermodynamics demonstrations comparing endothermic and exothermic salt dissolutions.

Dissolution of potassium chloride
KCl dissolution is mildly endothermic, slightly cooling the solution. It’s encountered in physiological and laboratory preparations and helps illustrate how different ionic salts give different heat effects.

Dissolution of ammonium chloride
Ammonium chloride absorbs heat when dissolved, often used historically in cold packs and classroom experiments to show endothermic dissolution and to teach about lattice versus solvation enthalpies.
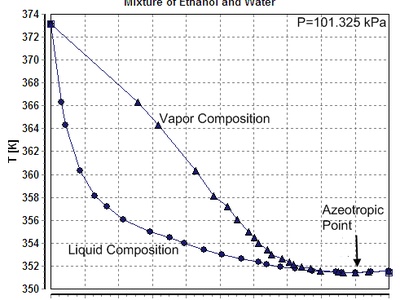
Vaporization of ethanol
Ethanol’s evaporation is endothermic, explaining evaporative cooling and rapid temperature drops when alcohols evaporate. It’s important in fuel vaporization, drying, and perfumery evaporation rates.

Dissociation of dinitrogen tetroxide
N2O4 dissociates into two NO2 molecules absorbing heat. This temperature-dependent equilibrium is a classic example of Le Chatelier’s principle and is visible as gas color changes with warming.

Formation of ozone
Ozone formation from oxygen is endothermic and driven by energetic UV photons or electrical discharges. It’s crucial for stratospheric UV shielding and in industrial/atmospheric chemistry where energy input creates O3.

Formation of nitric oxide
Making NO from N2 and O2 requires high temperatures and absorbs heat. It’s formed in combustion and lightning, central to atmospheric NOx chemistry and pollutant formation.
Electrolysis of water
Breaking water into hydrogen and oxygen is strongly endothermic and requires electrical energy. Electrolysis is fundamental for green hydrogen production and demonstrates conversion of electrical to chemical energy.

Photosynthesis (overall)
Photosynthesis is a large endothermic process driven by sunlight, storing solar energy in glucose bonds. It underpins the biosphere’s energy supply and is notable as a photochemical, not thermal, endothermic reaction.

Decomposition of calcium carbonate
Calcination of limestone to lime and carbon dioxide absorbs heat. This industrially important, strongly endothermic step is central to cement and lime manufacture and to CO2 release from geological materials.

Decomposition of ammonium chloride (to gases)
When heated, ammonium chloride decomposes into ammonia and hydrogen chloride gases, absorbing heat. It’s historically used in sublimation experiments and shows solid decomposition that produces gaseous products.

Thermal decomposition of sodium bicarbonate
Baking soda decomposes upon heating, absorbing heat and releasing CO2 and water vapor. This endothermic reaction is exploited in baking as a leavening step and in thermal decomposition studies.

Decomposition of magnesium carbonate
Magnesium carbonate thermally decomposes to magnesium oxide and carbon dioxide while absorbing heat. This endothermic carbonate decomposition is relevant in ceramics, metallurgy, and mineral thermal stability analysis.

Steam reforming of methane
Steam reforming is an industrially vital, strongly endothermic reaction converting methane and steam into syngas and hydrogen. It requires continuous heat input and is central to large-scale hydrogen and chemical feedstock production.

Decomposition of dinitrogen pentoxide
N2O5 decomposes endothermically to NO2 and O2 upon warming, relevant in nocturnal atmospheric chemistry and as an example of temperature-driven dissociation equilibria.