From solvents to pharmaceuticals, secondary alcohols are common players in both lab synthesis and everyday chemistry. They have one hydrogen on the carbon bearing the hydroxyl group, which gives them distinct reactivity for oxidation and substitution compared with primary or tertiary alcohols — a handy detail when planning reactions or interpreting spectra.
There are 16 Examples of Secondary Alcohols, ranging from 1-phenyl-1-propanol to sec-phenethyl alcohol. For each compound you’ll find below data organized with columns: IUPAC name or SMILES,Oxidation product (ketone),Boiling point (°C),Description (30-50 words); this layout makes it easy to compare structure, expected oxidation outcome, and practical properties you’ll find below.
How can I tell a secondary alcohol from primary or tertiary on a structure or in NMR?
Look at the carbon bonded to the OH: if it’s attached to two other carbons it’s secondary. In 1H NMR the OH proton may be broad or absent, but the neighboring CH proton(s) on a secondary carbon typically appear as multiplets; combined with carbon count and splitting, you can distinguish types.
What happens when a secondary alcohol is oxidized in the lab?
Oxidation of secondary alcohols typically yields a ketone (no further oxidation to carboxylic acids under normal conditions). Use reagents like PCC for mild oxidation or chromic reagents for more robust conditions, and expect retention of carbon skeleton with loss of the hydroxyl hydrogen.
Examples of Secondary Alcohols
| Common name | IUPAC name or SMILES | Oxidation product (ketone) | Boiling point (°C) | Description (30-50 words) |
|---|---|---|---|---|
| isopropanol | 2-propanol (CC(O)C) | acetone | 83°C | Isopropanol is a simple secondary alcohol with the OH on a carbon bonded to two carbons. It oxidizes to acetone under standard conditions. Common as a rubbing alcohol and solvent, notable for rapid evaporation and antiseptic use. |
| 2-butanol | 2-butanol (CC(O)CC) | 2-butanone (MEK) | 100°C | 2-Butanol is a straight-chain secondary alcohol (OH on a carbon bonded to two carbons). Oxidation yields 2-butanone, a common solvent. Found in small quantities in fermentation products and used as an intermediate in organic synthesis. |
| 2-pentanol | 2-pentanol (CC(O)CCC) | 2-pentanone | 119°C | 2-Pentanol is a secondary aliphatic alcohol with the hydroxyl-bearing carbon bonded to two carbons. It oxidizes to pentan-2-one. Used occasionally as a solvent and flavor intermediate, it’s a useful example of longer-chain secondary alcohol behavior. |
| cyclohexanol | cyclohexanol (C1CCCCC1O) | cyclohexanone | 161°C | Cyclohexanol is a cyclic secondary alcohol with the OH on a ring carbon bonded to two carbons. It oxidizes to cyclohexanone. Common in industry for nylon precursors and notable as a major bulk organic chemical intermediate. |
| cyclopentanol | cyclopentanol (C1CCCC1O) | cyclopentanone | 130°C | Cyclopentanol is a cyclic secondary alcohol (OH on a carbon bonded to two ring carbons). Oxidation gives cyclopentanone. Found in fragrances and as a small-molecule solvent, it illustrates ring-based secondary alcohol chemistry. |
| 1-phenylethanol | 1-Phenylethanol (C6H5CH(OH)CH3) | acetophenone | 203°C | 1-Phenylethanol is a benzylic secondary alcohol with OH on a carbon bonded to two carbons, including an aromatic ring. Oxidation yields acetophenone. Occurs in nature and fragrances; often used in stereochemistry examples and chiral synthesis. |
| benzhydrol | diphenylmethanol (Ph2CHOH) | benzophenone | 305°C | Benzhydrol (diphenylmethanol) is a benzylic secondary alcohol bonded to two phenyl groups. Oxidation produces benzophenone. Used as a reagent and intermediate in dye and pharmaceutical chemistry, notable for its diaryl-substituted secondary center. |
| 2,3-butanediol | 2,3-Butanediol (CH3CH(OH)CH(OH)CH3) | 2,3-Butanedione (diacetyl) | 180°C | 2,3-Butanediol is a vicinal diol with both OH groups on secondary carbons. Oxidation yields diacetyl (2,3-butanedione). It’s produced in fermentation, contributes to flavor compounds, and shows how multiple secondary OHs behave on oxidation. |
| propylene glycol | 1,2-Propanediol (CH3CH(OH)CH2OH) | hydroxyacetone (acetol) | 188°C | Propylene glycol is a diol with one secondary and one primary OH; the secondary OH is on a carbon bonded to two carbons. Partial oxidation gives hydroxyacetone. Widely used as antifreeze, food additive, and pharmaceutical solvent. |
| menthol | menthol (C10H19OH) | menthone | 212°C | Menthol is a naturally occurring cyclohexanol derivative with a secondary alcohol on a carbon bonded to two carbons. Oxidation yields menthone. Found in peppermint oil and widely used in flavorings, analgesic lotions, and cooling formulations. |
| isoborneol | isoborneol (bicyclic alcohol) | camphor | 212°C | Isoborneol is a bicyclic secondary alcohol with the OH on a carbon bonded to two carbons in a rigid framework. Oxidation yields camphor. It appears in essential oils and is notable for its use in classical organic transformations. |
| 2-ethylhexanol | 2-Ethylhexanol (CC(CC)CC(C)O) | 2-ethylhexanone | 184°C | 2-Ethylhexanol is a branched secondary alcohol used industrially as a plasticizer precursor and solvent. Oxidation produces 2-ethylhexanone. Its bulky chain gives low volatility for an alcohol and importance in polymer chemistry. |
| 2-hexanol | 2-hexanol (CCC(CO)CC) | 2-hexanone | 156°C | 2-Hexanol is a medium-chain secondary alcohol with the OH-bearing carbon bonded to two carbons. Oxidation yields hexan-2-one. Used as a solvent and reference compound in physical property studies, it demonstrates branched versus linear behavior. |
| glycerol (secondary center) | glycerol (1,2,3-propanetriol CH2OHCHOHCH2OH) | glycerone (dihydroxyacetone) or oxidized products | 290°C (decomposes) | Glycerol is a triol containing a central secondary OH (carbon bonded to two carbons). Oxidation of that center can produce dihydroxyacetone or further oxidized products. Common in biology and industry as a humectant and solvent. |
| 1-phenyl-1-propanol | 1-Phenyl-1-propanol (C6H5CH(OH)CH2CH3) | 1-Phenylpropan-1-one (propiophenone) | 204°C | 1-Phenyl-1-propanol is a benzylic secondary alcohol with the OH on a carbon bonded to two carbons (one aromatic). Oxidation yields propiophenone. Found in fragrance chemistry and useful in studying benzylic oxidation selectivity. |
| sec-phenethyl alcohol | 1-Phenyl-2-propanol (C6H5CH2CH(OH)CH3) | 1-Phenylpropan-2-one | 185°C | 1-Phenyl-2-propanol is a secondary alcohol where the OH-bearing carbon attaches to two carbons, one benzylic. Oxidation gives the corresponding ketone. It occurs as a fragrance intermediate and in studies of benzylic reactivity. |
Images and Descriptions

isopropanol
Isopropanol is a simple secondary alcohol with the OH on a carbon bonded to two carbons. It oxidizes to acetone under standard conditions. Common as a rubbing alcohol and solvent, notable for rapid evaporation and antiseptic use.

2-butanol
2-Butanol is a straight-chain secondary alcohol (OH on a carbon bonded to two carbons). Oxidation yields 2-butanone, a common solvent. Found in small quantities in fermentation products and used as an intermediate in organic synthesis.

2-pentanol
2-Pentanol is a secondary aliphatic alcohol with the hydroxyl-bearing carbon bonded to two carbons. It oxidizes to pentan-2-one. Used occasionally as a solvent and flavor intermediate, it’s a useful example of longer-chain secondary alcohol behavior.

cyclohexanol
Cyclohexanol is a cyclic secondary alcohol with the OH on a ring carbon bonded to two carbons. It oxidizes to cyclohexanone. Common in industry for nylon precursors and notable as a major bulk organic chemical intermediate.

cyclopentanol
Cyclopentanol is a cyclic secondary alcohol (OH on a carbon bonded to two ring carbons). Oxidation gives cyclopentanone. Found in fragrances and as a small-molecule solvent, it illustrates ring-based secondary alcohol chemistry.

1-phenylethanol
1-Phenylethanol is a benzylic secondary alcohol with OH on a carbon bonded to two carbons, including an aromatic ring. Oxidation yields acetophenone. Occurs in nature and fragrances; often used in stereochemistry examples and chiral synthesis.

benzhydrol
Benzhydrol (diphenylmethanol) is a benzylic secondary alcohol bonded to two phenyl groups. Oxidation produces benzophenone. Used as a reagent and intermediate in dye and pharmaceutical chemistry, notable for its diaryl-substituted secondary center.

2,3-butanediol
2,3-Butanediol is a vicinal diol with both OH groups on secondary carbons. Oxidation yields diacetyl (2,3-butanedione). It’s produced in fermentation, contributes to flavor compounds, and shows how multiple secondary OHs behave on oxidation.
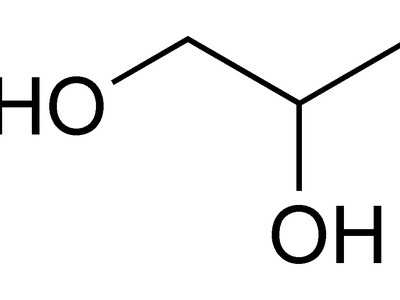
propylene glycol
Propylene glycol is a diol with one secondary and one primary OH; the secondary OH is on a carbon bonded to two carbons. Partial oxidation gives hydroxyacetone. Widely used as antifreeze, food additive, and pharmaceutical solvent.

menthol
Menthol is a naturally occurring cyclohexanol derivative with a secondary alcohol on a carbon bonded to two carbons. Oxidation yields menthone. Found in peppermint oil and widely used in flavorings, analgesic lotions, and cooling formulations.

isoborneol
Isoborneol is a bicyclic secondary alcohol with the OH on a carbon bonded to two carbons in a rigid framework. Oxidation yields camphor. It appears in essential oils and is notable for its use in classical organic transformations.

2-ethylhexanol
2-Ethylhexanol is a branched secondary alcohol used industrially as a plasticizer precursor and solvent. Oxidation produces 2-ethylhexanone. Its bulky chain gives low volatility for an alcohol and importance in polymer chemistry.

2-hexanol
2-Hexanol is a medium-chain secondary alcohol with the OH-bearing carbon bonded to two carbons. Oxidation yields hexan-2-one. Used as a solvent and reference compound in physical property studies, it demonstrates branched versus linear behavior.

glycerol (secondary center)
Glycerol is a triol containing a central secondary OH (carbon bonded to two carbons). Oxidation of that center can produce dihydroxyacetone or further oxidized products. Common in biology and industry as a humectant and solvent.

1-phenyl-1-propanol
1-Phenyl-1-propanol is a benzylic secondary alcohol with the OH on a carbon bonded to two carbons (one aromatic). Oxidation yields propiophenone. Found in fragrance chemistry and useful in studying benzylic oxidation selectivity.

sec-phenethyl alcohol
1-Phenyl-2-propanol is a secondary alcohol where the OH-bearing carbon attaches to two carbons, one benzylic. Oxidation gives the corresponding ketone. It occurs as a fragrance intermediate and in studies of benzylic reactivity.