In classrooms, labs and field demonstrations, decomposition reactions reveal how compounds break down into simpler substances — often releasing gases, heat or a change in color. They’re a staple when teaching reaction types because the signs are usually visible and straightforward to test.
There are exactly 25 Examples of Decomposition Reactions, ranging from Ammonium Dichromate to Sodium Bicarbonate. For each entry you’ll find below the Balanced equation, Reaction type, Conditions (°C, catalyst, light) so you can quickly compare thermal, catalytic and photochemical cases and pick examples suited to your needs.
How can I tell if a reaction is a decomposition reaction?
A decomposition reaction starts with a single reactant that breaks into two or more products; signs include gas evolution, color change, residue formation or measurable mass loss. Check stoichiometry for one reactant on the left and multiple products on the right, and confirm conditions (heat, catalyst, light) that typically drive the process.
What safety steps are essential for demoing these reactions?
Identify hazards for each example (toxic gases, high temperatures, explosive potential), use appropriate PPE and ventilation, perform small-scale trials, and have neutralizers or fire suppression ready; always follow institutional safety rules and review MSDS information before conducting demonstrations.
Examples of Decomposition Reactions
| Name | Balanced equation | Reaction type | Conditions (°C, catalyst, light) |
|---|---|---|---|
| Electrolysis of Water | 2H2O(l) -> 2H2(g) + O2(g) | Electrochemical | Electric current; requires an electrolyte (e.g., dilute H2SO4) |
| Hydrogen Peroxide (Catalytic) | 2H2O2(aq) -> 2H2O(l) + O2(g) | Catalytic | Catalyst (e.g., MnO2, KI) |
| Calcium Carbonate | CaCO3(s) -> CaO(s) + CO2(g) | Thermal | ~840-900°C |
| Sodium Bicarbonate | 2NaHCO3(s) -> Na2CO3(s) + H2O(g) + CO2(g) | Thermal | >50°C, common at baking temperatures (~180°C) |
| Potassium Chlorate | 2KClO3(s) -> 2KCl(s) + 3O2(g) | Thermal | ~400°C, or ~200°C with MnO2 catalyst |
| Sodium Azide | 2NaN3(s) -> 2Na(s) + 3N2(g) | Thermal | ~300°C, initiated by an electrical impulse |
| Ammonium Dichromate | (NH4)2Cr2O7(s) -> Cr2O3(s) + N2(g) + 4H2O(g) | Thermal | Ignition with heat |
| Silver Chloride | 2AgCl(s) -> 2Ag(s) + Cl2(g) | Photolytic | Sunlight or UV light |
| Electrolysis of Sodium Chloride | 2NaCl(l) -> 2Na(l) + Cl2(g) | Electrochemical | Electrolysis of molten salt at >801°C |
| Nitroglycerin | 4C3H5N3O9(l) -> 12CO2(g) + 10H2O(g) + 6N2(g) + O2(g) | Thermal | Shock, friction, or heat |
| Mercury(II) Oxide | 2HgO(s) -> 2Hg(l) + O2(g) | Thermal | ~500°C |
| Lead(II) Nitrate | 2Pb(NO3)2(s) -> 2PbO(s) + 4NO2(g) + O2(g) | Thermal | ~470°C |
| Copper(II) Carbonate | CuCO3(s) -> CuO(s) + CO2(g) | Thermal | ~190-290°C |
| Ozone | 2O3(g) -> 3O2(g) | Photolytic | UV light |
| Ammonium Nitrate | NH4NO3(s) -> N2O(g) + 2H2O(g) | Thermal | ~210-250°C |
| Hydrogen Peroxide (Biological) | 2H2O2(aq) -> 2H2O(l) + O2(g) | Biological | Catalase enzyme in cells |
| Electrolysis of Aluminum Oxide | 2Al2O3(l) -> 4Al(l) + 3O2(g) | Electrochemical | Electrolysis in molten cryolite at ~950°C |
| Carbonic Acid | H2CO3(aq) -> H2O(l) + CO2(g) | Spontaneous Chemical | Spontaneous in water; accelerated by enzymes |
| Glucose Fermentation | C6H12O6(aq) -> 2C2H5OH(aq) + 2CO2(g) | Biological | Yeast enzymes (zymase) in anaerobic conditions |
| Copper(II) Sulfate Pentahydrate | CuSO4·5H2O(s) -> CuSO4(s) + 5H2O(g) | Thermal | >150°C |
| Potassium Permanganate | 2KMnO4(s) -> K2MnO4(s) + MnO2(s) + O2(g) | Thermal | ~240°C |
| Nitrogen Triiodide | 2NI3(s) -> N2(g) + 3I2(g) | Thermal (contact) | Slight physical contact or vibration |
| Dinitrogen Tetroxide | N2O4(g) <=> 2NO2(g) | Thermal | Equilibrium is temperature-dependent; favors NO2 above 21°C |
| Phosphorus Pentachloride | PCl5(s) -> PCl3(l) + Cl2(g) | Thermal | >160°C |
| Formic Acid | HCOOH(l) -> H2O(l) + CO(g) | Catalytic | Dehydrating agent (e.g., concentrated H2SO4) |
Images and Descriptions
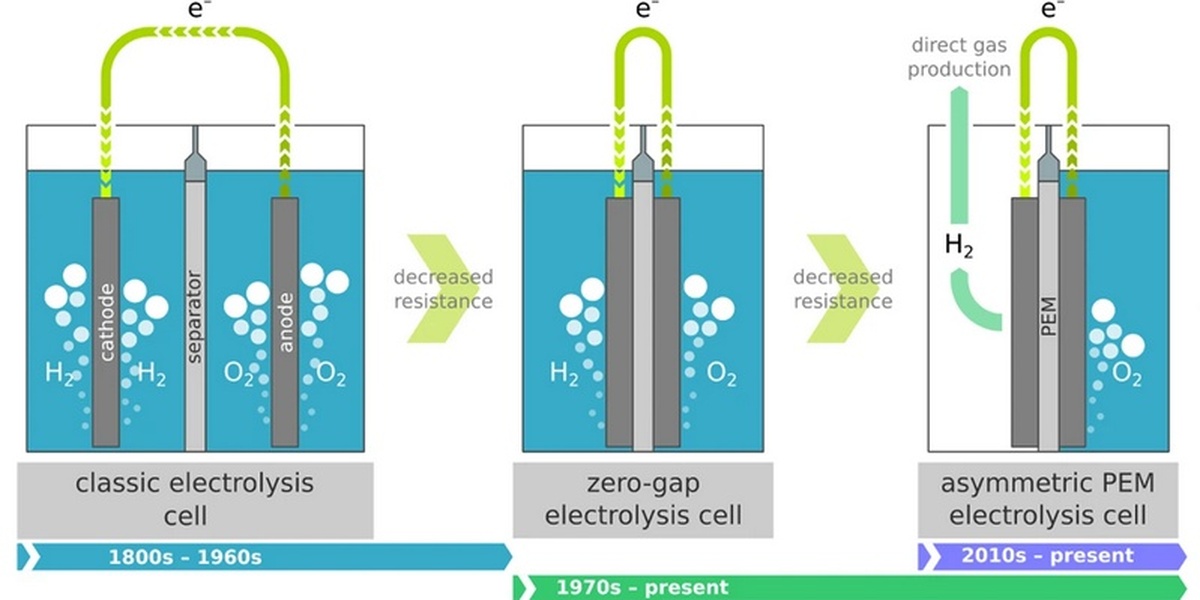
Electrolysis of Water
Passing an electric current through water splits it into hydrogen and oxygen gas. This is a key method for producing clean hydrogen fuel. The resulting gas mixture is highly flammable; avoid ignition sources.

Hydrogen Peroxide (Catalytic)
An inorganic catalyst rapidly breaks down hydrogen peroxide into water and oxygen. This reaction is famously used in the “elephant’s toothpaste” demonstration. Concentrated H2O2 is a strong oxidizer and can cause severe burns.

Calcium Carbonate
Heating limestone (calcium carbonate) produces quicklime and carbon dioxide. This process, called calcination, is a crucial step in manufacturing cement and steel. The high temperatures and CO2 release require industrial controls.
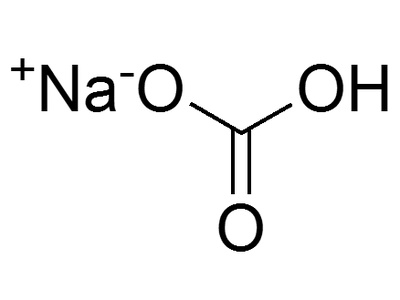
Sodium Bicarbonate
Baking soda decomposes with heat, releasing carbon dioxide gas bubbles that cause cakes and breads to rise. This safe and common reaction is essential for leavening in baked goods.
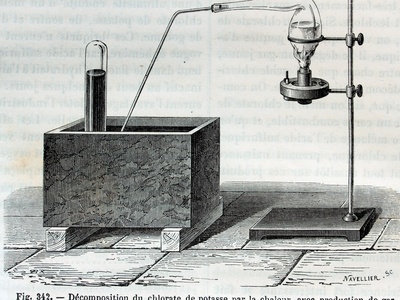
Potassium Chlorate
Heating potassium chlorate is a classic laboratory method for producing oxygen gas for experiments. It’s a powerful oxidizer used in some fireworks. The reaction can be explosive if contaminated; handle with extreme care.

Sodium Azide
This extremely rapid decomposition produces a large volume of nitrogen gas to inflate vehicle airbags during a collision. It is a life-saving application of a fast reaction. Sodium azide itself is highly toxic.

Ammonium Dichromate
This spectacular reaction, known as the “chemical volcano,” produces a cone of green chromium(III) oxide ash. It is a popular chemistry demonstration. Chromium compounds are toxic and carcinogenic; avoid inhalation.

Silver Chloride
Exposure to light causes this white solid to decompose into fine particles of black silver and chlorine gas. This reaction is the fundamental principle behind creating images on black-and-white photographic film.
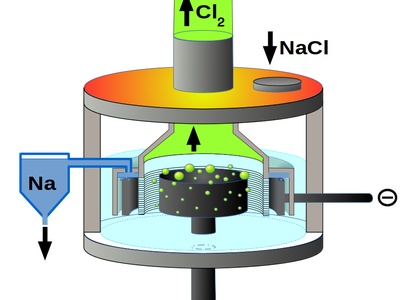
Electrolysis of Sodium Chloride
The Downs process uses an electric current to decompose molten table salt into sodium metal and chlorine gas. This is the primary industrial method for producing these two vital chemicals. Requires immense heat.

Nitroglycerin
This unstable liquid decomposes explosively, creating a massive volume of hot gas. It is the active ingredient in dynamite. It is extremely dangerous and highly sensitive to physical shock.

Mercury(II) Oxide
Heating this red-orange solid produces liquid mercury and oxygen gas. This historic experiment was crucial in Joseph Priestley’s and Antoine Lavoisier’s work on oxygen. Mercury and its compounds are highly toxic.

Lead(II) Nitrate
When heated, this white solid decomposes into yellow lead(II) oxide, oxygen, and toxic, brown nitrogen dioxide gas. This is a common decomposition example in chemistry labs. Lead compounds are toxic.

Copper(II) Carbonate
Heating this green powder causes it to transform into black copper(II) oxide while releasing carbon dioxide. This color change makes it a clear and simple demonstration of thermal decomposition.
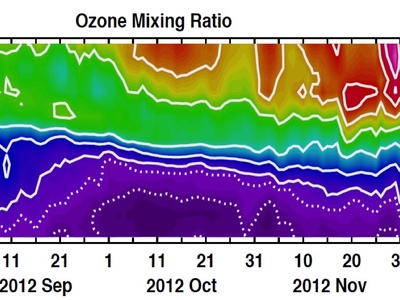
Ozone
In the stratosphere, high-energy UV light breaks ozone down into ordinary oxygen. This reaction is part of the natural ozone cycle that absorbs harmful UV radiation, protecting life on Earth.

Ammonium Nitrate
Careful heating of ammonium nitrate produces dinitrogen monoxide (laughing gas) and water. It is used as a fertilizer but can detonate violently if heated rapidly, making it dangerously explosive.
Hydrogen Peroxide (Biological)
The enzyme catalase, found in nearly all living organisms, safely breaks down toxic hydrogen peroxide into harmless water and oxygen. This is a vital process for protecting cells from oxidative damage.
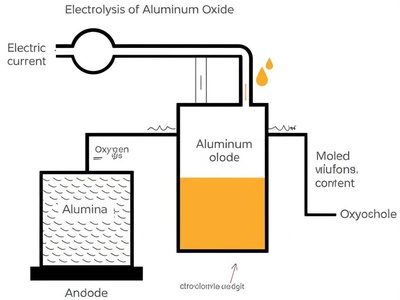
Electrolysis of Aluminum Oxide
The Hall-Héroult process uses massive amounts of electricity to decompose alumina (aluminum ore) into pure aluminum metal. This energy-intensive process is how virtually all commercial aluminum is produced.
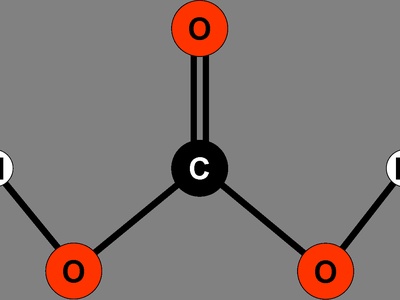
Carbonic Acid
Formed when CO2 dissolves in water, carbonic acid is unstable and quickly decomposes back into water and CO2. This is why carbonated drinks go flat and is critical for CO2 transport in the bloodstream.
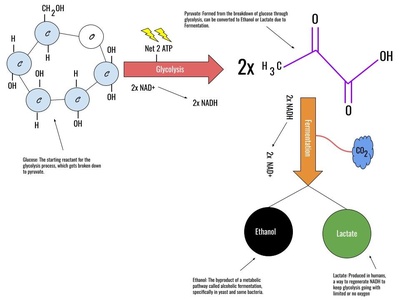
Glucose Fermentation
Yeast enzymes break down glucose into ethanol and carbon dioxide in the absence of oxygen. This ancient biochemical process is the basis for making alcoholic beverages and for the rising of bread dough.

Copper(II) Sulfate Pentahydrate
Heating the familiar blue crystals drives off the water molecules, leaving behind a white anhydrous powder. This reversible reaction is a classic lab demonstration of the decomposition of a hydrate.

Potassium Permanganate
Heating these deep purple crystals produces oxygen gas and other solid manganese compounds. It is another method for generating small amounts of oxygen in a laboratory setting. It is a very strong oxidizer.
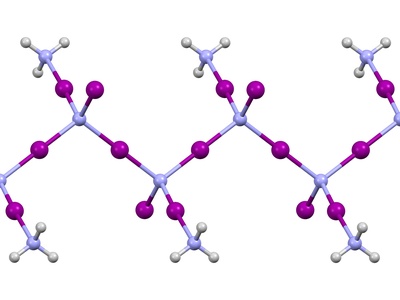
Nitrogen Triiodide
An extremely unstable contact explosive that decomposes with a loud snap and a cloud of purple iodine vapor at the slightest touch. It is a classic but highly hazardous demonstration. Never handle without expert supervision.

Dinitrogen Tetroxide
The colorless gas N2O4 decomposes into the reddish-brown, toxic gas NO2 as temperature increases. This reversible reaction is a striking visual demonstration of chemical equilibrium and Le Châtelier’s principle.

Phosphorus Pentachloride
Upon heating, solid phosphorus pentachloride decomposes into liquid phosphorus trichloride and chlorine gas. This reversible reaction is another common example used to teach chemical equilibrium in chemistry courses.

Formic Acid
A strong acid catalyst removes a water molecule from formic acid, leaving behind carbon monoxide gas. Carbon monoxide is a colorless, odorless, and highly toxic gas that must be handled in a fume hood.