From kitchens and classrooms to clinics and labs, knowing which cleaning agents to use — and how to use them safely — makes routine disinfection faster and more reliable. A concise reference helps teams pick the right product and follow required dwell times without guessing.
There are 15 Disinfectants, ranging from Accelerated hydrogen peroxide (AHP) to Sodium hypochlorite (bleach). For each entry you’ll find below Active ingredient,Common uses,Contact time (min) so you can compare efficacy, surface compatibility, and required contact times before use.
How do I choose the right disinfectant for different surfaces?
Pick a product based on the surface material, the type of microbes you need to control, and safety constraints (ventilation, residues, corrosion). Check manufacturer guidance for compatibility, follow label directions for concentration and contact time, and prefer EPA-registered or equivalent products for specific claims.
What should I know about contact times and real-world use?
Contact times vary widely—fast-acting alcohols often require under a minute on clean surfaces, while oxidizers and some quaternary ammonium formulations can need several minutes. Always clean visible soil first, apply the product at the labeled dilution, keep the surface wet for the full contact time, and consult the chart below for exact numbers.
Disinfectants
| Name | Active ingredient | Common uses | Contact time (min) |
|---|---|---|---|
| Sodium hypochlorite (bleach) | Sodium hypochlorite (NaOCl), typically 3–6% stock | Kitchens, bathrooms, hard nonporous surfaces, healthcare floors | 10 |
| Sodium dichloroisocyanurate (NaDCC) | Sodium dichloroisocyanurate — stabilized chlorine donor tablets | Portable disinfection, water treatment, healthcare surfaces, emergency kits | 10 |
| Chlorine dioxide | Chlorine dioxide gas or solution (ClO2) | Healthcare rooms, HVAC systems, food facilities, high‑level environmental decontamination | 5 |
| Hypochlorous acid (electrolyzed water) | Hypochlorous acid (HOCl), neutral pH chlorine species | Healthcare surfaces, food prep areas, infant/toddler surfaces | 1 |
| Ethanol / Isopropanol (alcohols) | Ethanol or isopropanol, typically 60–90% solutions | Small medical equipment, electronics, nonporous surfaces, labs | 1 |
| Quaternary ammonium compounds (quats) | Alkyl dimethyl benzyl ammonium salts (e.g., benzalkonium chloride) | Routine surface disinfection in households, hospitals, hospitality | 10 |
| Quat‑alcohol combinations | Quaternary ammonium salts plus ethanol or isopropanol | Healthcare surfaces, laboratories, wipe products, high‑touch areas | 5 |
| Hydrogen peroxide (standard) | Hydrogen peroxide, commonly 3–6% aqueous solutions | Food prep surfaces, labs, healthcare noncritical areas | 5 |
| Accelerated hydrogen peroxide (AHP) | Stabilized hydrogen peroxide formulations with surfactants and buffers | Healthcare surfaces, food service, veterinary clinics, wipes | 1 |
| Peracetic acid (peroxyacetic acid) | Peracetic acid (PAA), often combined with hydrogen peroxide | Food processing, endoscope reprocessing, wastewater, high‑level disinfection | 5 |
| Potassium peroxymonosulfate (oxidizing blend) | Potassium peroxymonosulfate with surfactants (e.g., Virkon) | Veterinary clinics, farms, laboratories, general surface disinfection | 10 |
| Phenolic compounds | Phenolic derivatives (o‑phenylphenol, cresols) | Hospital environmental surfaces, laboratory benches, medical equipment | 10 |
| Glutaraldehyde | Glutaraldehyde solutions, commonly 2% for high‑level disinfection | Medical instruments, endoscopes, lab equipment, immersion disinfection | 20 |
| Ortho‑phthalaldehyde (OPA) | Ortho‑phthalaldehyde solutions (~0.55%) | High‑level disinfection of flexible endoscopes and medical devices | 12 |
| Formaldehyde (formalin) | Formaldehyde gas or formalin solutions (formaldehyde in water) | Historic surface fumigation, lab decontamination, specialized sterilization | 30 |
Images and Descriptions

Sodium hypochlorite (bleach)
Bleach is a broad‑spectrum oxidizing disinfectant effective against bacteria, viruses and many fungi; higher concentrations needed for spores. Corrosive and irritating to eyes/airways, deactivated by organic matter; dilute per label, ventilate, and never mix with acids or ammonia.
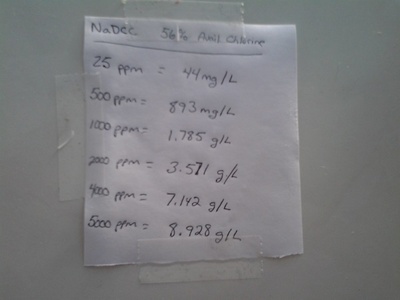
Sodium dichloroisocyanurate (NaDCC)
NaDCC releases chlorine when dissolved, providing a stable, easy‑to‑use source for surface and water disinfection. Effective against bacteria and viruses; corrosive to metals and irritating to skin/eyes; follow dilution and contact‑time instructions; useful where liquid bleach storage is impractical.
Chlorine dioxide
Chlorine dioxide is an effective oxidizing disinfectant and sterilant that penetrates biofilms and inactivates bacteria, viruses and spores. Requires controlled application, trained personnel, and monitoring—corrosive and respiratory irritant at high concentrations.

Hypochlorous acid (electrolyzed water)
HOCl is a mild, fast‑acting oxidizer produced by electrolysis or stabilized formulations; effective against bacteria and many viruses with low odor and low corrosivity. Short shelf life when dilute; follow label for concentration and avoid organic soiling.

Ethanol / Isopropanol (alcohols)
Alcohols rapidly inactivate many bacteria and enveloped viruses by protein denaturation; they evaporate quickly so surfaces must remain visibly wet for effectiveness. Flammable and can dry/crack plastics with repeated use; not sporicidal.

Quaternary ammonium compounds (quats)
Quats are surfactant disinfectants effective against bacteria and many enveloped viruses; variable activity vs. non‑enveloped viruses and spores. Low odor and corrosivity but can foam, leave residues, and cause skin/allergic reactions; efficacy reduced by organic matter.

Quat‑alcohol combinations
Combination products blend rapid alcohol kill with quat residual activity, improving spectrum and drying properties. Effective on many bacteria and viruses; flammable due to alcohol content, follow ventilation and label contact‑time guidance.

Hydrogen peroxide (standard)
Hydrogen peroxide is an oxidizing disinfectant active against bacteria, viruses and fungi; higher concentrations and longer contact times needed for some pathogens. Less corrosive than bleach but can bleach fabrics and irritate eyes/skin; decomposes to water and oxygen.

Accelerated hydrogen peroxide (AHP)
AHP combines peroxide with surfactants for faster action and broad spectrum activity including many viruses and bacteria. Short contact times and reduced corrosivity vs. plain peroxide; follow label, avoid mixing with incompatible chemicals.

Peracetic acid (peroxyacetic acid)
Peracetic acid is a potent oxidizer with rapid, broad spectrum activity including spores at certain concentrations. Effective at low temperatures but strongly corrosive and irritating; must be handled with ventilation, PPE, and proper dilution.

Potassium peroxymonosulfate (oxidizing blend)
This oxidizing formulation provides broad antimicrobial action including viruses and bacteria; often sold as powder to dissolve. Effective on many pathogens with moderate contact times; corrosive to some metals and irritating—use protective gear and rinse food contact surfaces.

Phenolic compounds
Phenolics disrupt cell walls and denature proteins; effective against many bacteria and some viruses. They persist as residues and can be toxic to aquatic life; handle with gloves and avoid use on food surfaces unless label permits.

Glutaraldehyde
Glutaraldehyde is a potent aldehyde high‑level disinfectant active against bacteria, fungi, viruses and some spores with adequate exposure. Toxic and irritating to skin, eyes and respiratory tract; requires controlled use, thorough rinsing of medical devices, and adequate ventilation.

Ortho‑phthalaldehyde (OPA)
OPA is a high‑level aldehyde disinfectant with broad activity and low vapor irritation compared with glutaraldehyde. Stains proteins (can discolor) and can cause allergic reactions; device rinsing per label is essential and unsuitable for porous surfaces.

Formaldehyde (formalin)
Formaldehyde is a highly effective biocidal aldehyde and fumigant that inactivates bacteria, spores and viruses. Carcinogenic and severely irritating; use is restricted, requires trained applicators, strict controls, and long aeration times before re‑entry.





