Oxidation sits at the heart of organic synthesis, converting functional groups and enabling strategic changes in complexity across medicinal, materials, and process chemistry. A concise, organized list of named and general oxidation methods makes it easier to compare selectivity, conditions, and likely outcomes when planning a route or troubleshooting a transformation.
There are 55 Oxidation Reactions, ranging from Aerobic alcohol oxidation (TEMPO/Cu) to Wacker oxidation. For each entry the data are organized as Substrate → Product,Reagents & conditions,Mechanism (brief) — you’ll find below.
How do I choose the right oxidation method for a specific substrate?
Pick a method by matching the desired chemoselectivity and oxidation level, checking functional-group tolerance, and considering catalyst or reagent availability; run small-scale screens (solvent, temperature, reagent equivalents) and consult examples with structurally similar substrates to predict outcomes.
What precautions should I take when scaling up an oxidation reaction?
Watch for exotherms, gas evolution, and oxidant decomposition: control addition rates, dilute if needed, monitor temperature closely, validate quench and workup procedures, and consider flow or semi-batch setups for safer heat and mass transfer before full-scale runs.
Oxidation Reactions
| Name | Substrate → Product | Reagents & conditions | Mechanism (brief) |
|---|---|---|---|
| PCC oxidation | 1° alcohol → aldehyde | PCC, CH2Cl2, 0–25 °C, ~1.1 equiv | Hydride transfer to Cr(VI), collapse to aldehyde |
| Jones oxidation | 1°/2° alcohol → carboxylic acid/ketone | CrO3,H2SO4,acetone,0–25 °C,excess Cr | Chromium(VI) oxidant performs successive hydride abstractions |
| Swern oxidation | 1°/2° alcohol → aldehyde/ketone | (COCl)2,DMSO,Et3N,−78→0 °C | Activated DMSO forms alkoxysulfonium, then elimination |
| Dess–Martin oxidation | 1°/2° alcohol → aldehyde/ketone | Dess–Martin periodinane, CH2Cl2,0–25 °C,1.1–2.0 equiv | O-activation by iodine(V), hydride transfer to iodine center |
| PDC oxidation | 1°/2° alcohol → aldehyde/ketone | PDC (pyridinium dichromate), CH2Cl2,0–25 °C | Cr(VI) mediated hydride abstraction from alcohol |
| Collins oxidation | 1°/2° alcohol → aldehyde/ketone | CrO3·2Py,CH2Cl2,0–25 °C | Cr(VI) complex performs hydride abstraction |
| TPAP (Ley) oxidation | 1°/2° alcohol → carbonyl | TPAP (0.05–0.1 equiv),NMO,CH2Cl2,room temp | Ru(VIII) oxo species mediate hydride transfer |
| TEMPO oxidation | 1° alcohol → aldehyde/carboxylic acid | TEMPO (cat),NaOCl/NaOBr or PhI(OAc)2,pH7–9,0–25 °C | Nitroxyl radical abstracts hydrogen, electron transfer to oxidant |
| MnO2 oxidation | allylic/benzylic alcohol → carbonyl | Activated MnO2, CH2Cl2,rt,stoichiometric | Surface hydride abstraction from activated alcohols |
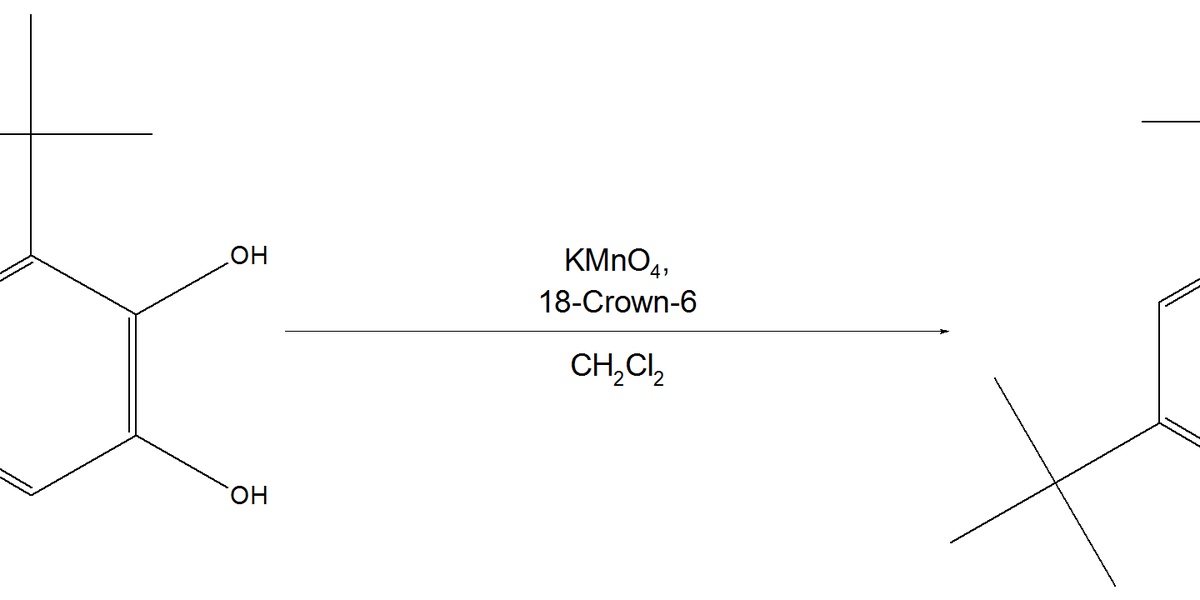
| KMnO4 oxidation | alkene/benzylic → diol/acid or cleavage products | KMnO4,aqueous or hot basic conditions,heat,excess | Multiple electron transfers yield diols or oxidative cleavage to carbonyls |
| Ozonolysis (oxidative workup) | alkene → carbonyls/carboxylic acids | O3,CH2Cl2 or MeOH,then H2O2 or O2 oxidative workup,−78→rt | O3 addition forms ozonide, oxidative workup converts to carbonyls/acids |
| Baeyer–Villiger oxidation | ketone → ester/lactone | mCPBA or peracid,H+,0–25 °C | Peracid inserts oxygen into carbonyl, rearrangement of R group |
| SeO2 allylic oxidation | allylic C–H → allylic alcohol | SeO2,t-BuOOH or dioxane,reflux | Selenium-mediated ene-type oxidation at allylic position |
| Wacker oxidation | terminal alkene → methyl ketone | PdCl2/CuCl2,H2O, O2 or air,40–80 °C | Pd(II) insertion, hydration, β-hydride elimination, reoxidation |
| Sharpless epoxidation | allylic alcohol → epoxide | t-BuOOH,Ti(OiPr)4,diethyl tartrate,−20→0 °C | Peroxide activated by Ti-tartrate delivers oxygen stereoselectively |
| Sharpless asymmetric dihydroxylation | alkene → vicinal diol | OsO4 (cat),NMO or K3Fe(CN)6,ligands,0–25 °C | OsO4 [3+2] cycloaddition, hydrolysis to diol |
| Epoxidation (peracid) | alkene → epoxide | mCPBA,CH2Cl2,0–25 °C,1.0–1.5 equiv | Concerted peracid oxygen transfer across the double bond |
| Periodic acid cleavage | vicinal diol → 2 carbonyls | HIO4 or NaIO4,aqueous solvent,rt | Diol chelates iodine, cleavage to carbonyl compounds |
| Pinnick oxidation | aldehyde → carboxylic acid | NaClO2,NaH2PO4,2-methyl-2-butene,0–25 °C | Chlorite oxidizes aldehyde to acid via chlorous intermediates |
| Tollens’ oxidation | aldehyde → carboxylate | Ag(NH3)2+,OH−,H2O,0–25 °C | Aldehyde donates electrons to Ag(I), forming Ag(0) mirror |
| Fehling’s/Benedict’s test | reducing sugar (aldehyde) → carboxylate | Cu2+ (alkaline tartrate),heat | Cu(II) reduced to Cu(I), aldehyde oxidized to acid |
| Kolbe electrolysis | carboxylate anion → radical → coupling products | Anodic oxidation in electrochemical cell,Pt anode,ether,rt | Anodic oxidation forms radical then decarboxylation and coupling |
| Glaser coupling (oxidative) | terminal alkyne → diyne | Cu(I) or Cu(II) salts,O2,amine solvent,rt–reflux | Copper oxidizes alkynyl anion, oxidative coupling forms C≡C–C≡C |
| Oxidative dearomatization (I(III)) | phenol → cyclohexadienone | PIDA or PIFA,MeOH/CH2Cl2,0–25 °C | Hypervalent iodine oxidizes phenol, electrophilic rearrangement forms dienone |
| Pfitzner–Moffatt oxidation | alcohol → aldehyde/ketone | DMSO,DCC,CH2Cl2,0–25 °C | Activated DMSO species abstracts hydride, collapses to carbonyl |
| DDQ dehydrogenation | allylic or benzylic substrate → dehydrogenated product | DDQ,benzene or CH2Cl2,rt–reflux | Single-electron/hydride abstraction yielding dehydrogenated product |
| N-oxidation of tertiary amines | tertiary amine → N-oxide | mCPBA or H2O2,CH2Cl2 or MeOH,0–25 °C | O transfer to nitrogen lone pair forming N–O bond |
| Sulfide oxidation | sulfide → sulfoxide → sulfone | mCPBA or H2O2,CH2Cl2 or MeOH,0–25 °C | Oxygen transfer to sulfur, stepwise oxidation to sulfone |
| Thiols oxidation | thiol → disulfide | I2,air,O2,or mild oxidants,pH 7–8,rt | Two thiols lose electrons forming disulfide bond |
| Fenton reaction (radical) | organic substrate → oxidized products | Fe2+,H2O2,acidic aqueous,0–80 °C | H2O2 activated by Fe2+ forms hydroxyl radical, abstracts H |
| Singlet oxygen (1O2) oxidation | alkene or diene → hydroperoxide/endoperoxide | Photosensitizer (e.g., Rose Bengal),O2,visible light,0–25 °C | Energy transfer forms 1O2, ene or [4+2] reactions add oxygen |
| Aerobic catalytic oxidation | alcohols or hydrocarbons → carbonyls/carboxylates | Pd,Cu,or TEMPO catalysts with O2 or air,rt–120 °C | Catalyst shuttles electrons from substrate to O2 reoxidant |
| Kharasch–Sosnovsky allylic oxidation | alkene C–H → allylic ester/alcohol | Cu(II) salts,peresters (e.g., t-BuOOAc),40–120 °C | Radical abstraction at allylic site, oxygenation via perester |
| Lead tetraacetate cleavage | 1,2-diol → carbonyl fragments | Pb(OAc)4,acetone or AcOH,0–25 °C | Lead(IV) forms complex, C–C cleavage to carbonyls |
| Permanganate oxidative cleavage (alkyne) | internal alkyne → carboxylic acids | KMnO4,hot aqueous,heat,excess oxidant | Oxidative cleavage at triple bond yields carboxylates |
| Oxidative phenol coupling | phenol → biaryl or diaryl ether | Cu(II),peroxide or O2,solvent,pH controlled,rt | Phenoxyl radicals couple to form C–C or C–O bonds |
| Enzymatic alcohol oxidation (ADH) | alcohol → aldehyde/ketone | Alcohol dehydrogenase,NAD+ or NADP+,physiological conditions | Hydride transfer from alcohol to NAD+, enzyme-catalyzed |
| Aldehyde dehydrogenase (ALDH) | aldehyde → carboxylic acid | ALDH,NAD+,H2O,physiological conditions | Hydride transfer to NAD+, hydration and oxidation to acid |
| Monoamine oxidase (MAO) | primary/secondary amine → imine → aldehyde/acid | MAO,FAD cofactor,O2,physiological conditions | Amine oxidation via flavin-mediated hydride transfer to O2 |
| Cytochrome P450 hydroxylation | C–H (various) → alcohol | P450,Fe(IV)=O active species,O2,NADPH,physiological temp | Oxygen insertion via high-valent iron-oxo species |
| Laccase oxidation | phenol → quinone or radical-coupled products | Laccase (Cu enzyme),O2,aqueous buffer,rt | Enzyme mediates one-electron oxidation via Cu centers |
| Glucose oxidase oxidation | β-D-glucose → glucono-δ-lactone | Glucose oxidase,O2,aqueous buffer,rt | Flavin-mediated two-electron oxidation of glucose to lactone |
| Combustion (complete) | hydrocarbon → CO2 + H2O | O2,high temperature,ignition source,combustion conditions | Rapid, multi-electron oxidation of carbon and hydrogen by oxygen |
| Atmospheric OH radical oxidation | volatile organic compound → oxidized organics | OH radical,O2,NOx,ambient temperature,photochemistry | H-atom abstraction or addition followed by O2 reaction yields oxidized products |
| SO2 atmospheric oxidation | SO2 → SO3 → sulfate | OH,O3,or metal-catalyzed pathways,aqueous aerosols | Radical or ozone-mediated oxidation converts sulfur dioxide to sulfur trioxide |
| NO oxidation to NO2 | NO → NO2 | O2 or O3,fast at ambient conditions,photochemical cycles | O2 or ozone oxidizes NO by electron transfer/ radical reaction |
| Autoxidation (radical chain) | alkane or lipid → hydroperoxide and oxidation products | O2,initiator or heat,ambient conditions,chain reaction | Radical chain propagation via peroxy radicals yields hydroperoxides and breakdown products |
| Aerobic alcohol oxidation (TEMPO/Cu) | alcohol → aldehyde/ketone | TEMPO,CuCl,NMI or base,O2,rt–60 °C | Catalyst mediates hydride transfer to TEMPO, O2 reoxidizes catalyst |
| Oxidative C–H functionalization (Pd-catalyzed) | C–H → C–O/C–N functionalized product | Pd(II) catalyst,oxidant (Cu,Ag,O2),solvent,heat | Pd inserts into C–H, functionalization with external oxidant regeneration |
| Oxidative cleavage of alkenes by OsO4/NaIO4 | alkene → carbonyls | OsO4 (cat),NaIO4,THF/H2O,0–25 °C | Dihydroxylation then periodic cleavage yields carbonyl fragments |
| Anodic oxidation (electrosynthesis) | various organic substrates → oxidized products | Electrochemical cell,appropriate electrodes,solvent,controlled potential | Electron removal at anode generates cationic or radical intermediates |
| Photocatalytic oxidation (Ru,Ir) | substrate → oxidized product | Ru(II) or Ir(III) photocatalyst,light,O2 or sacrificial acceptor,rt | Photoexcited catalyst promotes electron transfer to oxygen |
| Permanganate benzylic oxidation | benzylic C–H → carboxylic acid | KMnO4,aqueous,heat,alkaline conditions | Sequential hydrogen abstraction and oxygen insertion at benzylic carbon |
| Pummerer-type oxidative transformations | sulfide → α-oxidized products | Hypervalent iodine or peracids,acidic conditions,0–25 °C | Activation of sulfur followed by α-functionalization/oxidation |
| Peroxide epoxidation (Sharpless-Jung variant) | electron-rich alkene → epoxide | H2O2,ketone or acid catalyst,rt | Peroxide generates electrophilic oxygen species that add across alkene |
Images and Descriptions

PCC oxidation
PCC selectively oxidizes primary alcohols to aldehydes without overoxidation to carboxylic acids. Widely used in organic synthesis for sensitive substrates, mild conditions, and cases where aqueous workups and strong acids are undesirable.

Jones oxidation
Jones oxidation is a classical, strong chromium(VI)-based method that oxidizes primary alcohols to acids and secondary alcohols to ketones. It is robust but corrosive and generates toxic Cr waste, so modern alternatives are often preferred.

Swern oxidation
Swern oxidation converts alcohols to carbonyls under cold, mild, nonacidic conditions using activated DMSO. It is convenient for sensitive molecules because it avoids heavy metals and overoxidation, requiring low temperatures and careful handling of byproducts.

Dess–Martin oxidation
Dess–Martin periodinane (DMP) is a mild, selective reagent that oxidizes alcohols to carbonyls at ambient temperature. It is valued for low acidity, good chemoselectivity, and compatibility with sensitive functional groups.

PDC oxidation
PDC oxidizes alcohols to carbonyls similar to PCC but in slightly different conditions. It is used when chloride-sensitive substrates are present, though it still employs chromium(VI) and requires appropriate waste handling.

Collins oxidation
Collins reagent (chromium trioxide–pyridine) oxidizes alcohols to aldehydes or ketones under anhydrous conditions. It predates PCC but shares chromium-related toxicity concerns; used historically for sensitive dehydrative substrates.

TPAP (Ley) oxidation
TPAP (tetrapropylammonium perruthenate) catalyzes mild oxidation of alcohols to carbonyls with NMO reoxidant. It is catalytic, tolerant, and useful for sensitive molecules in small-scale synthesis.

TEMPO oxidation
TEMPO-mediated oxidations are selective for primary alcohols, often stopping at aldehydes or proceeding to acids depending on conditions. Widely used in green and catalytic aerobic oxidations of biomass and fine chemicals.

MnO2 oxidation
Activated manganese dioxide selectively oxidizes allylic and benzylic alcohols to aldehydes or ketones without touching many other functionalities, making it a classic reagent for selective oxidations.
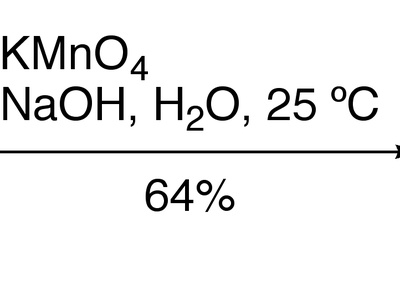
KMnO4 oxidation
Potassium permanganate is a strong, versatile oxidant for oxidizing alkenes, benzylic positions, and cleaving C=C bonds to carbonyls or carboxylic acids; it is robust but unselective under harsh conditions.

Ozonolysis (oxidative workup)
Ozonolysis cleaves alkenes to carbonyl fragments; with oxidative workup (H2O2) it gives carboxylic acids. It is powerful for pinpoint bond cleavage but requires careful handling of ozone and ozonides.

Baeyer–Villiger oxidation
Baeyer–Villiger oxidation converts ketones into esters or lactones via peracid-mediated oxygen insertion and rearrangement. It’s widely used in synthesis for ring expansion and for converting carbonyls to oxygenated products.

SeO2 allylic oxidation
Selenium dioxide selectively oxidizes allylic C–H bonds to allylic alcohols. Commonly used for introducing oxygen functionality adjacent to double bonds, though selenium toxicity and byproducts require careful workup.

Wacker oxidation
The Wacker process converts terminal alkenes to methyl ketones using Pd(II) catalysts and copper co-oxidants or oxygen. Industrially significant for large-scale oxidation of ethylene derivatives to ketones.

Sharpless epoxidation
Sharpless asymmetric epoxidation enables enantioselective epoxidation of allylic alcohols using a chiral titanium catalyst, a landmark in asymmetric synthesis for producing enantiomerically enriched epoxides.

Sharpless asymmetric dihydroxylation
Sharpless asymmetric dihydroxylation uses catalytic OsO4 with chiral ligands to introduce vicinal diols with high enantioselectivity, widely applied in complex molecule synthesis.

Epoxidation (peracid)
Peracid epoxidation is a straightforward oxidation converting alkenes to epoxides. Choice of peracid and conditions affords selectivity, and epoxides are versatile intermediates in synthesis.

Periodic acid cleavage
Periodic acid or sodium periodate cleaves vicinal diols oxidatively to aldehydes or ketones. This reaction is widely used in carbohydrate chemistry and for structural analysis of polyols.

Pinnick oxidation
Pinnick oxidation offers a mild, selective method to oxidize aldehydes to carboxylic acids without overoxidizing sensitive functionality. It tolerates many protecting groups and is common in total synthesis.

Tollens’ oxidation
Tollens’ reagent oxidizes aldehydes to carboxylates and deposits metallic silver, providing a classical qualitative test for aldehydes and a preparative oxidation method under mild, aqueous conditions.

Fehling’s/Benedict’s test
Fehling’s and Benedict’s reagents oxidize reducing sugars’ aldehyde functions to acids while reducing copper(II) to copper(I). These assays are important historically in carbohydrate analysis and clinical testing.

Kolbe electrolysis
Kolbe electrolysis oxidatively decarboxylates carboxylates at anode to give radicals that couple, useful for forming C–C bonds electrochemically without external oxidants other than electricity.

Glaser coupling (oxidative)
Glaser oxidative coupling joins terminal alkynes to form diynes using copper and oxygen. This oxidative homocoupling is a classical method for building conjugated alkyne linkages.

Oxidative dearomatization (I(III))
Hypervalent iodine reagents oxidatively convert phenols into cyclohexadienone-type products, enabling ring functionalizations and cascade reactions in complex molecule synthesis.

Pfitzner–Moffatt oxidation
Pfitzner–Moffatt is a DMSO-based method for oxidizing alcohols to carbonyls using DCC as activator. It is milder than chromium methods and useful where low acidity and compatibility matter.

DDQ dehydrogenation
DDQ (2,3-dichloro-5,6-dicyanobenzoquinone) is a strong dehydrogenative oxidant used to remove H2 (dehydrogenation) and aromatize or oxidize electron-rich substrates in synthesis.

N-oxidation of tertiary amines
Oxidation of tertiary amines to N-oxides with peracids is a useful transformation to alter basicity and reactivity, commonly used in medicinal chemistry and functional group interconversions.

Sulfide oxidation
Oxidation of sulfides to sulfoxides and further to sulfones is straightforward using peroxides or peracids. Control of equivalents enables selective formation of sulfoxide or full oxidation to sulfone for diverse synthetic needs.

Thiols oxidation
Thiols oxidize to disulfides under mild oxidants or air, a biologically important process in protein folding and a practical synthetic way to make disulfide-linked molecules and protect thiol functionality.

Fenton reaction (radical)
The Fenton reaction generates hydroxyl radicals from hydrogen peroxide and ferrous iron; these radicals rapidly oxidize organic molecules. It is used in wastewater treatment and radical-mediated organic transformations.

Singlet oxygen (1O2) oxidation
Singlet oxygen photooxygenation oxidizes alkenes and dienes to hydroperoxides or endoperoxides. Important in natural product synthesis and biological oxidative stress studies.

Aerobic catalytic oxidation
Aerobic oxidations use molecular oxygen as terminal oxidant in catalytic systems to oxidize alcohols and hydrocarbons. They are attractive for green chemistry and industrial oxidations when safe and selective.

Kharasch–Sosnovsky allylic oxidation
Kharasch–Sosnovsky reactions perform allylic oxidations via copper-catalyzed radical pathways, introducing oxygen functionality adjacent to double bonds, useful in complex molecule construction.

Lead tetraacetate cleavage
Lead tetraacetate oxidatively cleaves vicinal diols to carbonyl fragments and is used for selective bond disconnections, though toxicity limits its modern use in favor of safer reagents.

Permanganate oxidative cleavage (alkyne)
Strong permanganate oxidation cleaves alkynes to carboxylic acids and is a powerful way to break multiple bonds and introduce oxidation at terminal or internal triple bonds.
Oxidative phenol coupling
Oxidative coupling of phenols using copper or peroxidase enzymes forms biaryl or diaryl ether linkages, a key step in natural product biosynthesis and lignin formation.

Enzymatic alcohol oxidation (ADH)
Alcohol dehydrogenases oxidize alcohols biologically via hydride transfer to NAD+, crucial in metabolism and widely exploited for stereoselective transformations in biocatalysis.

Aldehyde dehydrogenase (ALDH)
Aldehyde dehydrogenases oxidize toxic aldehydes to carboxylic acids in metabolism, using NAD+ as acceptor. They are central to detoxification and drug metabolism pathways.

Monoamine oxidase (MAO)
Monoamine oxidases oxidatively deaminate neurotransmitters and drugs via FAD-dependent mechanisms, producing imines that hydrolyze to aldehydes; important in pharmacology and neurochemistry.

Cytochrome P450 hydroxylation
Cytochrome P450 enzymes catalyze regio- and stereoselective oxygenation of C–H bonds in metabolism and synthesis, inserting oxygen into hydrocarbons using molecular oxygen and NADPH.

Laccase oxidation
Laccases use oxygen to oxidize phenolic substrates to quinones or radicals, enabling crosslinking, polymerization, and biotechnological applications in bioremediation and biosensors.

Glucose oxidase oxidation
Glucose oxidase is used in biosensors and food preservation, converting glucose to gluconolactone while reducing oxygen, providing a reliable enzymatic oxidation for analytical chemistry.

Combustion (complete)
Combustion is the rapid oxidation of hydrocarbons to carbon dioxide and water, releasing energy. It underpins engines, power generation, and flames, but also contributes to pollution and CO2 emissions.

Atmospheric OH radical oxidation
Hydroxyl radical-initiated oxidation is the atmosphere’s primary cleanup mechanism for VOCs, driving formation of oxygenated species, secondary organic aerosol, and influencing air quality and climate.
SO2 atmospheric oxidation
Atmospheric oxidation of SO2 to sulfate particles leads to acid rain and aerosol formation; gas-phase and aqueous-phase pathways both contribute to environmental sulfur cycling.

NO oxidation to NO2
Oxidation of nitric oxide to nitrogen dioxide is central to atmospheric chemistry and urban smog formation, affecting air quality and acting as precursor to ozone and nitric acid.

Autoxidation (radical chain)
Autoxidation is the spontaneous oxygen-driven radical oxidation of hydrocarbons and lipids, responsible for fuel degradation, polymer aging, and food rancidity, driven by O2 and radicals.

Aerobic alcohol oxidation (TEMPO/Cu)
TEMPO/Cu aerobic systems oxidize alcohols using oxygen as terminal oxidant, offering greener alternatives to stoichiometric oxidants with good selectivity and scalability.

Oxidative C–H functionalization (Pd-catalyzed)
Palladium-catalyzed oxidative C–H functionalization converts C–H bonds into C–O or C–N bonds using external oxidants for catalyst reoxidation, enabling direct late-stage modifications in complex molecules.

Oxidative cleavage of alkenes by OsO4/NaIO4
A two-step oxidative sequence uses OsO4-catalyzed dihydroxylation followed by NaIO4 cleavage to convert alkenes to carbonyl compounds. It is a stereospecific and predictable bond-cleavage strategy.

Anodic oxidation (electrosynthesis)
Anodic oxidation uses electricity to remove electrons from substrates, enabling reagent-free oxidation pathways for coupling, dehydrogenation, and functionalization under green electrochemical conditions.

Photocatalytic oxidation (Ru,Ir)
Photocatalytic oxidations harness light-activated catalysts to oxidize substrates via electron transfer to oxygen or other acceptors, enabling mild, selective transformations with temporal control.

Permanganate benzylic oxidation
Strong permanganate oxidizes benzylic positions to carboxylic acids, a reliable way to convert alkyl aromatics into aromatic acids for analytical and synthetic applications.

Pummerer-type oxidative transformations
Pummerer-like oxidative methods enable α-functionalization adjacent to sulfur via oxidation and rearrangement. They are useful for constructing complex sulfur-containing motifs and elaborating sulfide substrates.

Peroxide epoxidation (Sharpless-Jung variant)
Peroxide-based epoxidations are economical oxidations of electron-rich alkenes to epoxides, used industrially and in lab synthesis when milder or less expensive oxidants than peracids are preferred.