From lab benches to nutrition labels, the small molecules that build proteins are everywhere in research and everyday life. A quick familiarity with their names and basic properties makes it easier to read sequences, plan meals, or follow biochemical discussions.
There are 22 Amino Acids, ranging from Alanine to Valine. For each entry I list the 3-letter code, 1-letter code, and Side chain class; you’ll find below.
How do I use the 3-letter and 1-letter codes when reading sequences?
Use the 1-letter code for compact sequence notation (for example, Alanine = Ala = A) and the 3-letter code when you need clarity in structural or lab notes. The 1-letter system speeds up sequence work, while the 3-letter form and the side chain class help you infer chemical behavior like polarity or charge.
Which residues are essential and why does that matter?
Essential residues are those the body can’t synthesize and must obtain from the diet; knowing which ones are essential helps with nutrition planning and understanding metabolic limits. When you scan the list below, the essential subset highlights dietary needs and can explain why some proteins or supplements matter more in certain diets.
Amino Acids
| Name | 3-letter code | 1-letter code | Side chain class |
|---|---|---|---|
| Alanine | Ala | A | Nonpolar aliphatic |
| Arginine | Arg | R | Basic |
| Asparagine | Asn | N | Polar uncharged |
| Aspartic acid | Asp | D | Acidic |
| Cysteine | Cys | C | Sulfur-containing |
| Glutamine | Gln | Q | Polar uncharged |
| Glutamic acid | Glu | E | Acidic |
| Glycine | Gly | G | Special |
| Histidine | His | H | Basic |
| Isoleucine | Ile | I | Nonpolar aliphatic |
| Leucine | Leu | L | Nonpolar aliphatic |
| Lysine | Lys | K | Basic |
| Methionine | Met | M | Sulfur-containing |
| Phenylalanine | Phe | F | Aromatic |
| Proline | Pro | P | Special |
| Serine | Ser | S | Polar uncharged |
| Threonine | Thr | T | Polar uncharged |
| Tryptophan | Trp | W | Aromatic |
| Tyrosine | Tyr | Y | Aromatic |
| Valine | Val | V | Nonpolar aliphatic |
| Selenocysteine | Sec | U | Selenium-containing |
| Pyrrolysine | Pyl | O | Special |
Images and Descriptions
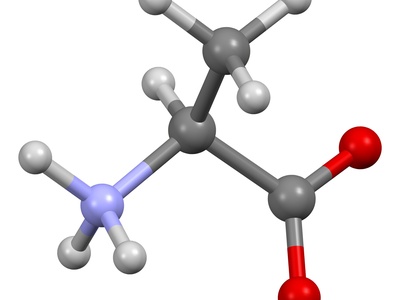
Alanine
Alanine is a small, nonpolar amino acid common in proteins and muscle tissue. It helps stabilize protein structure and can be used for energy in metabolism. Notably, its simple methyl side chain makes it structurally unreactive.
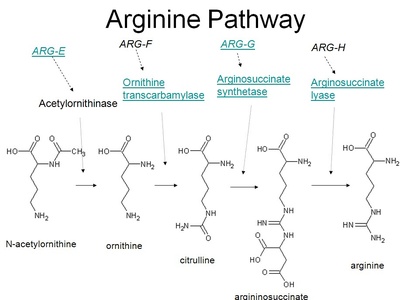
Arginine
Arginine is a positively charged basic amino acid found in many proteins and concentrated in the urea cycle and nitric oxide production. It supports cell signaling and vasodilation by serving as a precursor to nitric oxide, important for blood flow regulation.

Asparagine
Asparagine is a polar, uncharged amino acid often located on protein surfaces and in secreted glycoproteins. It stabilizes structure through hydrogen bonding and participates in protein recognition; notable for its role in N-linked glycosylation motifs.
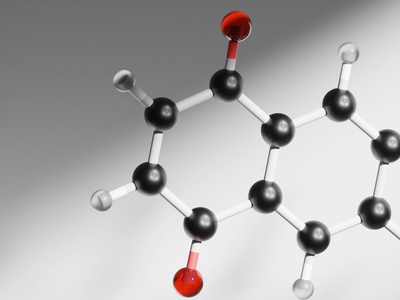
Aspartic acid
Aspartic acid is a negatively charged acidic amino acid common in active sites and membrane proteins. It participates in catalysis, metal binding and charge interactions. Notable for frequently acting as a proton donor or ligand in enzyme mechanisms.

Cysteine
Cysteine is a sulfur-containing amino acid found in active sites and structural disulfide bonds of secreted and extracellular proteins. Its thiol side chain forms disulfide bridges that stabilize tertiary structure and serves in redox chemistry and metal coordination.

Glutamine
Glutamine is a polar, uncharged amino acid abundant in blood and rapidly dividing cells. It donates nitrogen in biosynthesis, supports immune and intestinal health, and serves as a carbon and nitrogen carrier; notable for high concentrations in muscle.
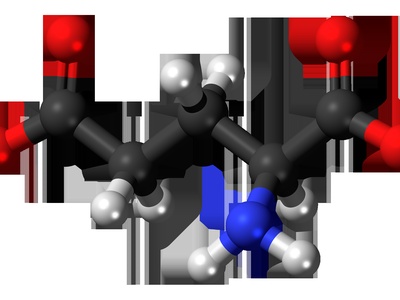
Glutamic acid
Glutamic acid is a negatively charged amino acid abundant in brain and dietary proteins. It acts as a major excitatory neurotransmitter precursor and participates in nitrogen metabolism. Notable for its role in synaptic signaling and as monosodium glutamate flavor enhancer.

Glycine
Glycine is the smallest amino acid, achiral and flexible, found in collagen and many proteins. It provides structural flexibility and fits in tight spaces, critical for collagen’s triple helix and central in neurotransmission as an inhibitory transmitter in the spinal cord.
Histidine
Histidine is a basic amino acid with an imidazole side chain important in enzyme active sites and metal binding. It helps buffer physiological pH and participates in catalysis. Notable for versatile protonation states near physiological pH.

Isoleucine
Isoleucine is a branched-chain, nonpolar amino acid concentrated in muscle proteins. It contributes to hydrophobic cores, supports energy production during exercise and is essential in diet. Notable as one of three branched-chain amino acids important for muscle metabolism.

Leucine
Leucine is a branched, hydrophobic amino acid common in structural and metabolic proteins. It stimulates muscle protein synthesis and acts as an important energy source during fasting. Notable for its regulatory role in the mTOR growth-signaling pathway.
Lysine
Lysine is a basic, essential amino acid abundant in many dietary proteins and important in collagen crosslinking. It participates in protein interactions, acetylation sites, and immune function. Notable for its dietary importance and role in viral protein binding.

Methionine
Methionine is a sulfur-containing, essential amino acid that often initiates protein synthesis and donates methyl groups via S-adenosylmethionine. It’s found in dietary proteins and enzymes. Notable for its role as the start codon’s encoded amino acid in most organisms.
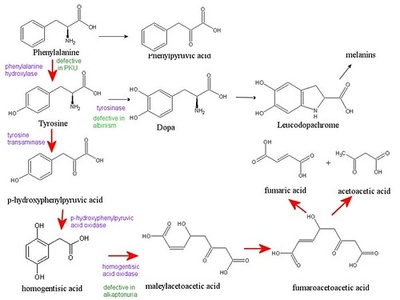
Phenylalanine
Phenylalanine is an aromatic, essential amino acid present in dietary proteins and many enzymes. It provides hydrophobic packing and serves as a precursor to tyrosine and neurotransmitters. Notable for its link to phenylketonuria when metabolism is impaired.

Proline
Proline is a cyclic imino acid that imposes kinks in polypeptide chains and is abundant in collagen and structural proteins. Its rigid ring affects folding and stability. Notable for disrupting alpha helices and stabilizing collagen’s unique conformation.

Serine
Serine is a polar, uncharged amino acid commonly found at active sites, in signaling proteins and on protein surfaces. Its hydroxyl side chain is a frequent site for phosphorylation and catalytic activity. Notable for roles in enzyme mechanisms and regulation.
Threonine
Threonine is a polar, essential amino acid with a hydroxyl-containing side chain important in structural and signaling roles. It is commonly phosphorylated in regulatory proteins and contributes to protein stability. Notable as an essential dietary amino acid.

Tryptophan
Tryptophan is a large aromatic amino acid found in many proteins and dietary sources. It’s a precursor to serotonin and niacin and influences protein fluorescence. Notable for its rarity and strong absorbance useful in spectroscopic protein studies.

Tyrosine
Tyrosine is a polar aromatic amino acid present in many enzymes and signaling proteins. Its phenolic OH can be phosphorylated in signaling pathways and it serves as a precursor for catecholamines and thyroid hormones. Notable for regulatory phosphorylation roles.

Valine
Valine is a branched, hydrophobic amino acid abundant in muscle and dietary proteins. It supports protein structure, energy metabolism and is essential in diet. Notable as one of the three branched-chain amino acids important for muscle function.

Selenocysteine
Selenocysteine is the selenium-containing amino acid incorporated into selenoproteins via UGA recoding. Found in enzymes like glutathione peroxidases, it participates in redox catalysis and antioxidant defense. Notable for replacing sulfur with selenium for enhanced reactivity.

Pyrrolysine
Pyrrolysine is a rare, proteinogenic amino acid found in certain methanogenic archaea and bacteria. It is inserted at UAG codons via a dedicated tRNA and appears in methyltransferases involved in methylamine metabolism. Notable for expanding the genetic code in select organisms.






