In 1949 Willard Libby described a method to measure carbon-14 in organic materials, a breakthrough that reshaped archaeology and earned him the Nobel Prize in Chemistry in 1960.
But despite its power, radiocarbon dating is often misunderstood. Persistent myths about radiocarbon dating lead to misread ages, false controversies (think the Shroud of Turin) and misplaced distrust of archaeological and climate records.
This piece debunks eight common misconceptions and explains what 14C ages really mean, when results are reliable, and what limits matter. The myths are grouped into four themes: fundamentals, contamination and preservation, calibration and precision, and scope and misuse. Concrete examples include the 1988 Shroud study, Ötzi the Iceman, and the IntCal20 calibration curve.
Fundamentals: How radiocarbon dating works (and what it doesn’t do)

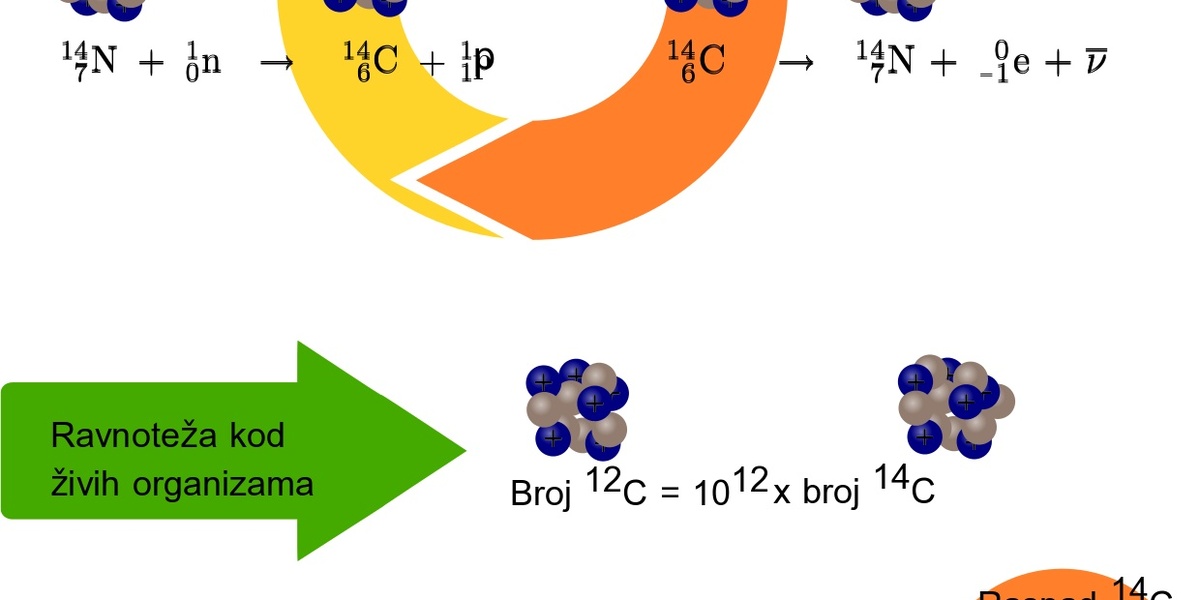
Radiocarbon dating rests on a simple chain: cosmic rays produce carbon-14 in the upper atmosphere, that radioactive isotope mixes into atmospheric CO2, plants fix it via photosynthesis, and animals acquire it by eating plants. When an organism dies it stops exchanging carbon, and the 14C decays with a half-life of about 5,730 years (±30 years).
Laboratories measure remaining 14C and report a radiocarbon age in 14C years. That measurement must be converted to calendar years using calibration curves derived from tree rings, corals, varves and other archives—modern standard curves are bundled in the IntCal series (IntCal20 was released in 2020).
Real-world limits matter: 14C dating is effective up to roughly 50,000 years, after which the remaining 14C is at or below background and uncertainty rises quickly. Also, not every sample type or context is equally datable.
1. Myth: Radiocarbon dating gives exact calendar years
Many expect a single-year answer like “1543 CE.” In reality a lab returns a radiocarbon age (in 14C years) plus a measurement uncertainty. That number is translated to calendar years by calibration curves because atmospheric 14C has varied through time.
Calibration is empirical: tree-ring chronologies give annual resolution for many intervals, while other records fill gaps. Calibration can shift a radiocarbon age by decades or even centuries in some intervals. The 1988 radiocarbon tests on the Shroud of Turin, for example, yielded a calibrated range of about 1260–1390 CE at standard probabilities.
Laboratories normally report probability ranges (often 1σ and 2σ). Archaeologists quote calibrated ranges with probabilities—for example, 1840–1780 BCE at 95% probability—so single-year certainty is rare and almost always unjustified.
2. Myth: Radiocarbon dating works the same for every material and age
Not all materials behave equally. Wood, charcoal, seeds, bone, shell and peat have different chemistry and preservation pathways. Pretreatment protocols (acid-base-acid for charcoal, collagen extraction for bone) remove contaminants that would otherwise bias results.
Modern accelerator mass spectrometry (AMS) lets labs measure tiny samples—micrograms to milligrams—but small samples still require careful pretreatment and context. Reservoir effects mean marine or some freshwater organisms can appear hundreds of years older than contemporaneous terrestrial samples unless corrections are applied.
And there’s the “old wood” problem: a charcoal sample dates the death of the tree tissue, which may be centuries before it was used by people. For people-sized samples, examples like Ötzi the Iceman (~3300 BCE) show careful sample choice and multiple lines of evidence yield reliable dates.
Contamination and preservation: Why context and chemistry matter
Contamination and preservation drive many misunderstandings. Modern carbon introduced by handling or conservation will skew an age younger; conservation glues or older carbon sources can skew ages older. Labs use chemistry and quality metrics to spot and deal with these issues.
3. Myth: Any contamination ruins a radiocarbon date beyond usefulness
Contamination is real, but not always catastrophic. Labs test for contamination and apply pretreatments to remove it. For bone, the atomic C:N ratio in collagen (typically 2.9–3.6) is a standard quality indicator; values outside that range flag problems.
How much contamination matters depends on its proportion. A little modern contamination can shift an age by hundreds of years; a lot can swamp the signal. That’s why archaeologists prefer dating short-lived materials (seeds, annual plants) or multiple, independent samples and sometimes send material to more than one lab to cross-check results.
In practice a careful lab report will list pretreatment steps and QA numbers so users can judge reliability rather than assuming a single value is unusable after any contamination event.
4. Myth: Museum conservation always prevents accurate radiocarbon testing
Museums often worry that past treatments make dating impossible. Many consolidation or adhesive treatments can be removed, or labs can target uncontaminated fragments or internal samples. AMS’s small-sample needs make minimally invasive sampling feasible in many cases.
That said, sampling decisions balance preservation against scientific value. Some objects are too fragile or culturally sensitive to sample. Best practice is early consultation between conservators and radiocarbon labs, full documentation of treatments, and sampling only where the scientific gain justifies the risk.
Numerous successful studies used tiny removed fragments for AMS dating; others declined to sample because risks outweighed benefits. It’s a pragmatic, case-by-case decision.
Calibration, precision, and interpretation: The limits of certainty
Calibration turns measured 14C ages into calendar probability ranges using independent records: tree rings, corals, speleothems and varves feed into IntCal. The curve isn’t smooth; wiggles and plateaus mean the same radiocarbon age can map to several calendar ranges or a broader span in some intervals.
IntCal20 (2020) is the current widely used calibration dataset, and it’s updated as new measurements improve resolution. That empirical basis is why calibrated results are more trustworthy than a raw 14C number alone.
5. Myth: Calibration makes radiocarbon results subjective or “adjusted” to fit a narrative
Calibration is not an ad-hoc adjustment. It’s an objective translation based on independently dated archives; the data and software (calibration programs) are publicly available. You input a measured 14C age and its uncertainty; the software outputs a reproducible probability distribution against the calibration curve.
Updates like IntCal20 refine the curve as new tree-ring, coral or varve data arrive. Techniques such as wiggle-matching—fitting sequences of tree rings or timber rings to the calibration curve—use the pattern of ring-by-ring variation to sharpen dates, not to force results into a preferred story.
6. Myth: A smaller margin of error always means a better or more trustworthy date
Tighter lab precision (a smaller ± in 14C years) is desirable, but calendar precision also depends on where that 14C age falls on the calibration curve. A ±20 14C-year result might map to a narrow calendar window in one interval and a broad, multi-century window in another because of curve shape.
Always read the lab report: it should include the raw 14C age, the lab uncertainty and the calibrated ranges (1σ and 2σ). Both the laboratory precision and the calibration context determine how confidently you can place a sample in time.
Scope and misuse: Popular controversies and where radiocarbon dating is misrepresented

Public controversies and sensational headlines often arise from misunderstanding what 14C dating can and cannot do. Misuse includes overclaiming precision, ignoring sample context, or cherry-picking dates to support a prior narrative.
Good practice combines radiocarbon with other methods—dendrochronology, stratigraphy, typology or inscriptions—so that dates are interpreted within a broader evidential framework.
7. Myth: Radiocarbon dating can settle any historical argument on its own
Radiocarbon dates the dated material, not the historical event people often want to time. Timber reused in later construction, old wool in a burial, or residual charcoal in a hearth can mislead if context is ignored.
Robust conclusions come from multiple lines of evidence. Dendrochronology can give annual precision for wood, stratigraphy ties samples to occupation layers, and typology or inscriptions provide cultural context. Radiocarbon is powerful, but it rarely stands alone as the final word.
8. Myth: Disputed high-profile examples show radiocarbon is “wrong” or untrustworthy
High-profile disputes—like the Shroud of Turin—often turn on sampling choices, unrecognized contamination or incomplete documentation, not on a failure of the 14C method itself. The 1988 Shroud tests returned a calibrated range around 1260–1390 CE, and critics pointed to sampling and textile heterogeneity as reasons to question that single result.
Such controversies drive better practice: clearer chain-of-custody, multiple laboratories, and more careful sampling schemes. Re-sampling or complementary analyses have clarified several early contentious results, reinforcing the method when properly applied.
Summary
- Radiocarbon ages must be calibrated to produce calendar probability ranges; calibration curves (e.g., IntCal20) are empirical and publicly documented.
- Sample type, pretreatment and archaeological context critically affect reliability; reservoir effects and “old wood” can bias apparent ages.
- Contamination is manageable: labs use pretreatment and QA metrics (for example, collagen C:N ≈ 2.9–3.6) and dating multiple samples improves confidence.
- Calibration complexity and occasional high-profile disputes reflect sampling and interpretation issues, not a fundamental flaw in the method.
- When you see a radiocarbon claim, ask for the calibrated ranges, the raw 14C age and uncertainty, pretreatment details and whether short-lived or multiple samples were used.