In 1875, Santiago Ramón y Cajal published detailed drawings of neurons that changed how scientists understood the brain’s structure.
Brain science matters because the clinical burden is enormous: nearly 1 billion people worldwide live with neurological or mental-health conditions, and about 50 million people live with dementia.
Neuroscience is entering a decade where improved imaging, computation, and ethics-aware deployment will reshape medicine, technology, and society.
This piece lays out exactly eight concrete trends to watch across three areas: Clinical & medical advances, Technology & data, and Society & research infrastructure.
Read on to see how these developments will affect patients, labs, companies, and policy makers.
Clinical and Medical Advances
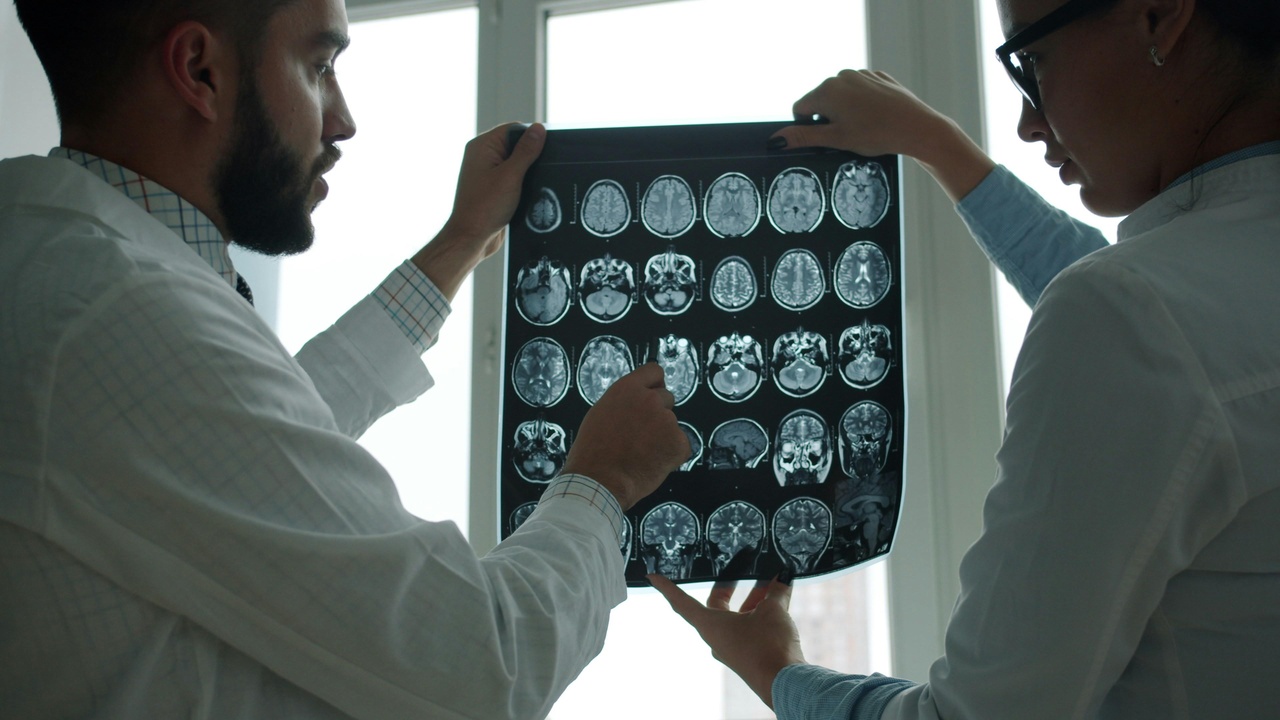
Clinical need—Alzheimer’s, stroke, epilepsy—plus converging technologies are forcing faster translation from bench to bedside.
About 50 million people with dementia and rising stroke incidence worldwide mean the next 5–15 years will focus on diagnostics, devices, and targeted therapies.
Below are three trends likely to change patient care in the near term.
1. Precision neuromedicine: genetics and biomarkers guide treatment
Genomic data and molecular biomarkers will let clinicians tailor treatments for epilepsy, Parkinson’s, Alzheimer’s, and some psychiatric disorders.
Large-scale genomic studies and consortia (for example, ADNI) have expanded variant catalogs and phenotypes. CSF amyloid and tau assays matured in the 2010s, and blood-based amyloid/tau biomarkers saw major advances after 2018, shortening diagnostic timelines.
The consequence is practical: faster, more accurate diagnosis; genetics-informed therapy selection in epilepsy; and better patient stratification that can reduce clinical-trial failure rates.
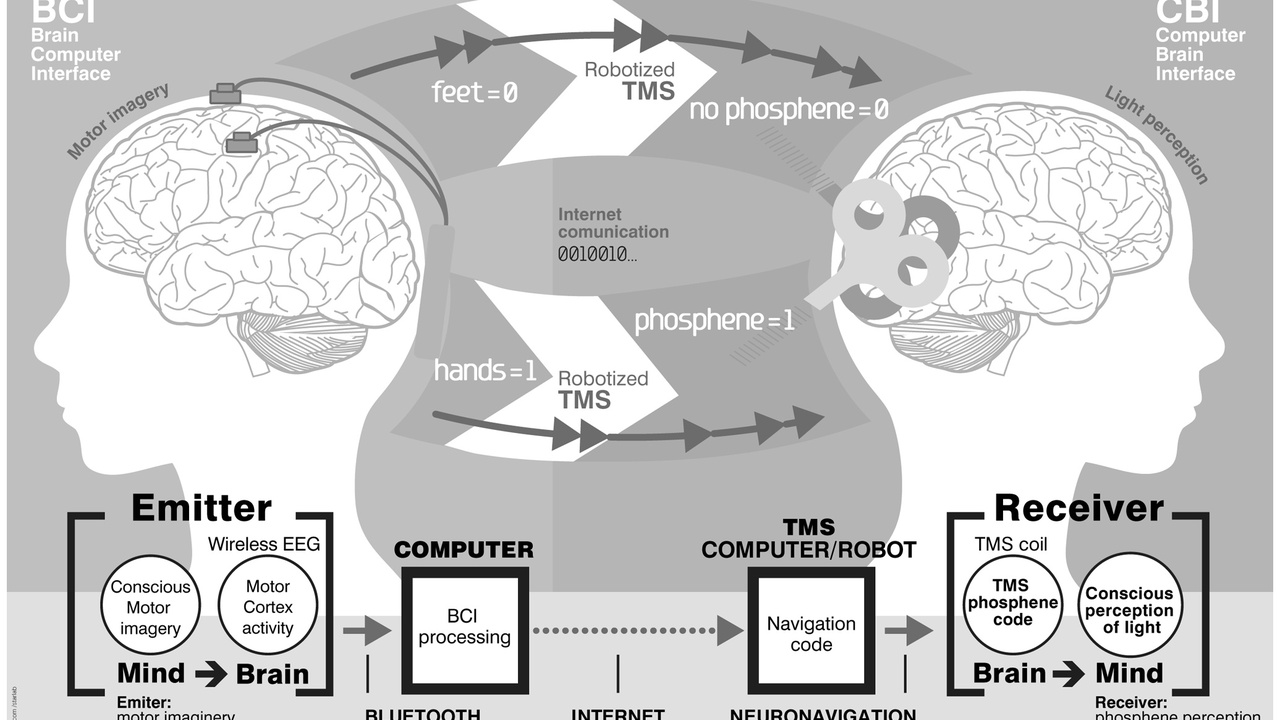
2. Neuroprosthetics and brain–computer interfaces move from lab to daily life
Intracortical BCIs and advanced prosthetic limbs are moving from demonstrations to real-world assistive tools for people with paralysis and limb loss.
Clinical trials since the early 2010s have shown intracortical devices enabling typing, cursor control, and robotic-arm manipulation. Dozens of human participants have received implantable interfaces in the past decade, and companies such as Neuralink, Blackrock Neurotech, and Synchron are scaling implant and clinical programs.
Everyday impact includes restored communication, independent device control, and improved quality of life, though durability, infection risk, and regulatory pathways remain hurdles to broad adoption.
3. Next-generation neuroimaging: higher resolution and functional insight
Advances in MRI, MEG, and optical methods are shifting imaging from coarse maps to circuit-level insight in humans.
Ultra-high-field MRI (7T and beyond), which became widely available to research centers in the 2010s, can yield roughly 1 mm anatomical resolution for many contrasts. MEG and high-density EEG provide millisecond timing for functional dynamics. Large projects such as the Human Connectome Project (launched 2009) created protocols and datasets that accelerated these methods.
Clinically, higher resolution imaging improves surgical planning, enables earlier disease detection, and supplies richer biomarkers for trials.
Technology, Data, and Computation
Artificial intelligence, expanding datasets, and novel hardware will accelerate discovery and create new product classes.
Growing investments and cross-disciplinary teams are scaling projects that link models, data, and devices.
Here are three computational and data trends to watch.
4. AI and computational neuroscience: models that predict and explain
Machine learning is shifting from pattern-finding to more interpretable models that predict treatment response and circuit behavior.
Deep convolutional and recurrent networks applied to fMRI and electrophysiology produced key papers in the late 2010s and early 2020s linking artificial networks to cortical responses. Researchers now use ML for automated lesion detection, EEG-based seizure prediction, and trial enrichment.
These approaches deliver measurable accuracy gains in research studies and can shorten time to diagnosis or improve patient selection for trials. Expect models to become tools clinicians use for prognosis and simulated intervention planning.
5. Connectomics and big data: mapping circuits at scale
The goal is wiring diagrams across scales, from synapses to whole-brain networks, built from large coordinated datasets.
Major efforts such as the Human Connectome Project (since 2009) and the BRAIN Initiative (launched 2013) have driven collection standards and tooling. Connectomic datasets now include thousands of human imaging datasets and, in model organisms, millions of synapses reconstructed at EM scale.
Applications include revealing disease-related circuit changes, guiding neurosurgical targets, and informing brain-inspired computing, though storage, annotation, and automated segmentation remain technical bottlenecks.
6. Neuromorphic computing and edge neurotech
Hardware that emulates neuronal computation will enable low-power on-device processing for wearables and implants.
Prototypes such as IBM TrueNorth and Intel Loihi appeared in the 2010s and have matured into research platforms in the 2020s. Commercial interest is rising for on-chip inference to reduce latency and power draw.
Expected impacts include battery-friendly neural prosthetics, real-time seizure detection on wearables, and privacy-preserving local analytics. Adoption will depend on programming models and integration with mainstream ML toolchains.
Society, Policy, and Research Infrastructure

Technical progress raises social questions and infrastructure choices that will determine who benefits from advances.
Below are two trends that will shape governance, access, and workforce needs.
7. Neuroethics, privacy, and regulation become central
As devices access neural signals, legal and ethical questions about privacy, consent, and cognitive liberty move from academic debate to policy making.
Concerns include ownership of neural data, informed consent for implanted devices, and the potential for misuse in surveillance or coercion. Professional groups and standards bodies (including IEEE and national bioethics councils) increased public discussions and regulatory engagement in the 2020s.
Practically, companies and clinicians will need clear compliance pathways, and patients will demand transparent consent language and data controls.
8. Open science, infrastructure, and workforce growth
Open datasets, standard formats, and collaborative platforms speed discovery and broaden participation.
Repositories such as OpenNeuro and Neurodata Without Borders, plus large public initiatives like the Human Connectome Project and the BRAIN Initiative, provide data that lowers barriers to entry.
Demand for neuroengineers, data scientists, and clinician‑scientists will grow. Institutions should invest in interdisciplinary graduate programs and shared data infrastructure to scale training and ensure equitable access.
Summary
- Precision neuromedicine and biomarkers will speed diagnosis and enable targeted therapies, reducing trial failure and improving outcomes.
- Implantable and wearable neuroprosthetics, supported by improved imaging and neuromorphic hardware, will bring device-assisted communication and mobility into daily life.
- AI, connectomics, and large public datasets will accelerate hypothesis testing and enable predictive, interpretable models that inform clinical decisions.
- Policy, neuroethics, and open science will determine who benefits—about 50 million people with dementia underscore the urgency—so follow open-data repositories and standards efforts and support interdisciplinary training.